Cross-Topic Integration (7 of 26)
Understanding Gibbs free energy is crucial to the understanding of bioenergetics. To understand the meaning of free energy change, a good place to start is with the Carnot cycle. The Carnot cycle is the archetypal, theoretical model of a reversible engin. It shows in the simplest way possible how it is that a spontaneous process, such as heat flowing from a hot body to a cold body, can be coupled to make some other nonspontaneous process occur. This can happen up to the point where you don't begin to actually decrease the entropy of the universe. You need both the first and second laws of thermodynamics in your analytical toolkit to understand the Carnot cycle. Each stage of the Carnot cycle should be thought through in terms of both laws.
The first law of thermodynamics allows us to describe each step in terms of heat flow, work performed, and internal energy change. The second law gives us the tools to understand how the changes in entropy in the universe are in balance thorughout every stage. Every stage of the Carnot cycle is microscopically reversible, and the entropy of the universe does not increase.
By being reversible, the Carnot cycle finds the limit of heat flow that can be turned into work without decreasing the entropy of the universe. In this sense, the Carnot cycle represents the ideal (most efficient) thermodynamic cycle possible. Compared to the Carnot cycle, all real cycles would result in even more heat being lost to the cold sink for a given amount of work.
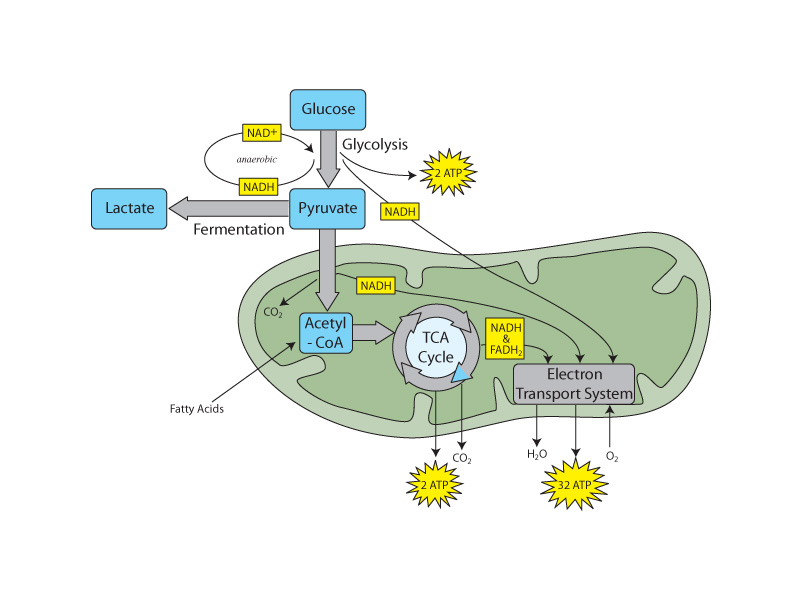
The Gibbs free energy is a state function of a chemical system that keeps track of how changes in the system affect the entropy of the universe. A change that increases the entropy of the universe is reflected as a negative free energy change. Just like in a Carnot cycle, a spontaneous process, such as combustion of glucose, can be coupled with a nonspontaneous process, such as ATP synthesis up to the point where the overall free energy change would become positive. Applied to chemical thermodynamics, the Carnot cycle is a model for chemical equilibrium. If the overall free energy change is zero, the coupled system is in equilibrium, just like the Carnot cycle. If the overall free energy change is still negative, such as in oxidative metabolism, the process proceeds forward spontaneously.